Lots of interesting things to find when you turn over the rocks of Hansen et al 2006. These are comments on work in progress, but, to say the least, there appear to be some curious decisions and methodologies.
Reviewing briefly, the key figure in Hansen et al 2006 is its Figure 5 comparing recent SST measurements in the Pacific Warm Pool to Mg/Ca reconstructions.

Figure 1. Original Caption: Hansen et al 2006 Fig. 5. Modern sea surface temperatures (5, 6) in the WEP compared with paleoclimate proxy data (28). Modern data are the 5-year running mean, while the paleoclimate data has a resolution of the order of 1,000 years.
Now I don’t have any objection to focussing on the Western Equatorial Pacific. In fact, I think that there’s much to be said for focussing on a region that’s both extremely important and with relatively uniform temperature. It’s how it’s done that concerns me. In order to derive the above SST reconstruction, a transfer function relating Mg/Ca to SST is required. The transfer function used in Hansen et al 2006 is taken from Medina-Elizalde and Lea 2005 and is as follows:
(1) SST (deg C) = ln (Mg.Ca(m)/ 0.3) / 0.089 or equivalently
(2) Mg.Ca = 0.3 exp [ 0.089* SST]
Medine-Elizalde and Lea 2005 (see Legend to Medina-Elizalde and Lea Figure 2) cited Lea et al 2000 as authority for this transfer function. Now Lea et al 2000 is a relatively early paper in Mg/Ca paleo-calculations. It noted, but did not adjust for, preferential dissolution of Mg at depth in the Ontong Java Plateau (OJP); they noted that failure to account for such dissolution would result in colder paleo-temperature estimates. A more complicated formula with an adjustment method for dissolution was presented in Dekens et al 2002 (including Lea). However, for some reason, the updated formula was not applied in either Medina-Elizalde and Lea 2005 or Hansen et al 2006. Had the Dekens et al formula been used, the reconstruction would have been a little bit warmer as shown in Figure 2 below. (The 29.2 deg C benchmark is taken from Dekens et al 2002). I show this figure with a big asterisk as it seems to me that there’s an elementary algebraic error in the calculation of the formula in Dekens et al 2002 – which I will present for discussion below.
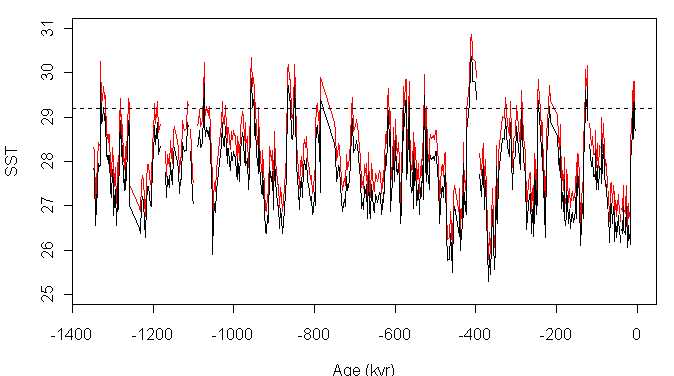
Figure 2. Re-stated Paleo-reconstruction using formula of Dekens et al 2002 – red.
Update Oct. 3 I wrote to David Lea asking why the older formula was used and he replied:
Hi Steve: the adjustment based on the Dekens 2002 equation is quite small at the location and depth of Hole 806b (~0.3 deg in SST), and, in the absence of time-varying dissolution corrections, only serves to change the absolute SST value. For that reason we chose to maintain the earlier calibration to be consistent with the published data in the 2000 study.
Sincerely, David
End Update
However, there’s a lot more than this going on, which I’ll try to summarize here.
Lea et al 2000
Let’s start with Lea et al 2000, beginning with their key Figure 2.
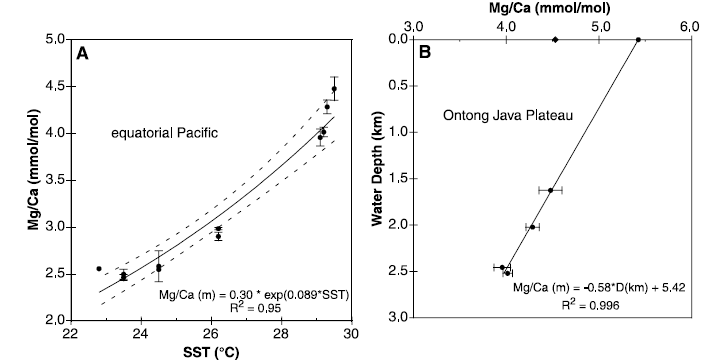
Figure 3. Lea et al 2000 Fig. 2. (A) Pacific core-top G. ruber calibration for Mg/Ca versus mean annual SST (43). Each point is the average of two or more analyses. The range of core-top water depths is 1625 to 3200 m. The standard error of the exponential fit is 60.6¡C. Dashed lines indicate the 95% conàÆà
⽤ence intervals for the curve fit.(B) Mg/Ca in G. ruber shells from core-tops on the Ontong Java Plateau as a function of water depth. The àÆà
⽬led diamond at 0 m is the Mg/Ca value for a plankton tow sample taken at SST 5 27.2¡C off southwest Puerto Rico. The filled circle at 0 m is the same sample corrected for the 2 deg C warmer temperature of the WEP. The data indicate that shell Mg/Ca decreases by about 12% per 1 km increase in water depth. See text for details.
The left panel shows the derivation of their calibration equation – the one used above. The right panel shows the effect of increasing core-top depth on dissolution of Mg/Ca with the effect on foraminifera shells reported by the authors as being visible. The points used to calibrate the left panel equation come from two locations – the “warmer” cores are from the Western Equatorial Pool (WEP) including core ODP806B; the “cooler” cores are from the Eastern Equatorial Pacific, all around the Galapagos. The core top Mg/Ca measurements are all dated through radiocarbon and dates of 4000-6000 BP are assigned to the core top measurements. The SST measurements are modern SST measurements from Levitus 1994.
Think about what is illustrated here (and it took me a long time to understand it): the left panel shows a regression relationship between Holocene Optimum G. ruber Mg/Ca and modern SST!?! At most, this could illustrate a general geographic relationship but it can’t be used to calibrate anything, especially if it’s supposed to be calibration within a degree for the Framework Convention. But it’s worse. The right hand values from the WEP are “net” Mg/Ca values after dissolution. So the regression relationship is between partially dissolved Holocene Optimum foraminifera Mg/Ca and modern SST, with no accounting for the degree of dissolution!?!
Now there has been considerable discussion in the specialist literature about dissolution. I’ve spent a couple of days trying to replicate the above diagram in order to test the effect of applying different approachs to allowing for dissolution. The cores used in the above diagram appear almost certainly to be (from left to right) the following 11 cores: TR163-32; TR163-27; TR163-28; TR163-20B;TR163-22; TR163-18; TR163-19; ODP806B; MW91-9 08; MW91-9 34; MW91-9 38. (See Data Digression below). Figure 4 below shows my emulation of Lea et al Figure 2, using data from various publications – see Data Data Digression.

Figure 4. Emulation of Lea et al 2000 Figure 2.
The right panel indicates the presumed “original” Mg/Ca ratios prior to dissolution en route to sediments. (The dissolution occurs strongly at depth and Lea mentions that Mg is completely dissolved in OJP cores at 3500 m and below. The dissolution pertains to pCO2 values at depth. BTW there is some discussion about changing pCO2 values at depth over time in this literature which should be considered by people interested in carbon cycle and depp ocean exchange.) The “original” Mg/Ca values at surface, that gave rise to the WEP core top values, are approximately 5.42, a indicated by the surface intercept of the line on the right panel.
Figure 5 below shows these adjusted values in red in the same format as Lea et al Figure 2 (together with one modern measurement from the Atlantic in orange). Obviously the “adjusted” Mg/Ca values don’t fit the Lea et al 2000 curve at all. (BTW it’s my impression that the foraminifera in the EEP were not dissolved to the same extent as the OJP samples and adjustment is not as important there, but it’s something that needs to be verified.) So where does this leave us? The transfer equation in Lea et al 2000, used in Medina-Elizalde and Lea 2005 and Hansen et al 2006, appears to be completely inappropriate and without any logical basis, let alone statistical basis. So let’s go back to Dekens et al 2002 and see how they handled dissolution and see what can be salvaged.
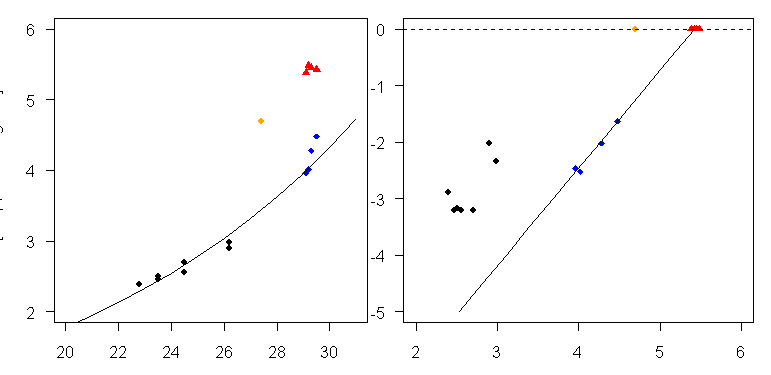
Figure 5. Mg/Ca values adjusted for dissolution shown in red in same format as Figure 3.
Dekens et al 2002
It’s hard to see why the formula in Dekens et al 2002 wasn’t used in the later studies rather than the primitive methodology of Lea et al 2000. As noted above, perhaps it was because the reported adjustment in Dekens et al 2002 didn’t appear to be very large – but it’s still large enough to make a difference especially if we’re talking about half a degree or so. The actual expression in Dekens et al 2002 was:
(3) 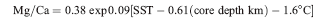
which expressed in the same form as (1) is obviously:
(4) SST = ln ( Mg.Ca/0.38) +0.61*km +1.6
Dekens et al 2002 state, but do not derive this formula. In order to develop a rational transfer function, one obviously has to get away from regressing partially-dissolved Holocene Optimum core tops against modern SST and it appears that this was attempted in Dekens et al, but with some odd algebra.
Barker et al 2005 have a recent and sensible survey of Mg/Ca proxy calibration based on modern sediment trap data for calibration resulting in the following formula:
(5) Mg/Ca = 0.38 exp [ 0.09* SST]
This formula is shown in their Figure 2 as shown below:
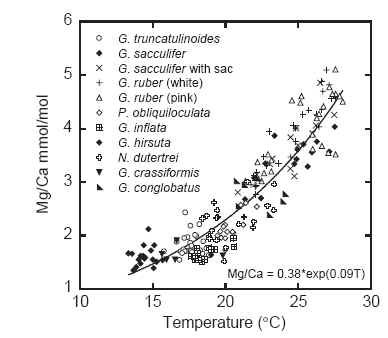
Figure 5. Barker et al 2005 Fig. 2. Mg/Ca calibration results of Anand et al. (2003) for several species of planktonic foraminifera. Temperatures shown here are the isotopically derived calcification temperatures of Anand et al. (2003). A single temperature equation may be used to describe all data àÆà°r ¼ 0:93àÆà
⼺ Modified after Anand et al. (2003).
Notice that the key outside parameters of the Barker et al transfer function are identical to the corresponding parameters used in Dekens et al 2002: the “preexponent” parameter of 0.38 occurs in both as does the inside parameter of 0.09. So it’s evident that Dekens et al 2002 applied the relationship later articulated in Barker et al 2005, the relationship presumably available in the community in 2002. Dekens et al appear to have used this relationship on reconstructed “original” Mg/Ca values, after allowing for dissolution.
Now the adjustment for dissolution from the right panel of Lea et al 2000 (with denoting the surface (original) Mg/Ca values prior to dissolution – the value appropriate for the Barker et al 2005 equation.
(6)
The parameter 0.58 for the depth relationship is obviously very close to the figure of 0.61 in the Dekens et al 2002 equation; I will apply this parameter in the following calculations in place of the value of 0.58. Thus:
(6a)
Substituting this relationship into (5) and using baby steps, we get:
(7)
(8)
This is similar in appearance, but materially different from the formula of Dekens et al 2002:
Equation (8) follows logically from the literature, but the Dekens et al 2002 equation only appears possible by dropping a bracket or some other error in the simple algebra.
So what happens if formula (8) is applied to actual data – doing the algebra in baby steps:
(9)
gives the following reconstruction:
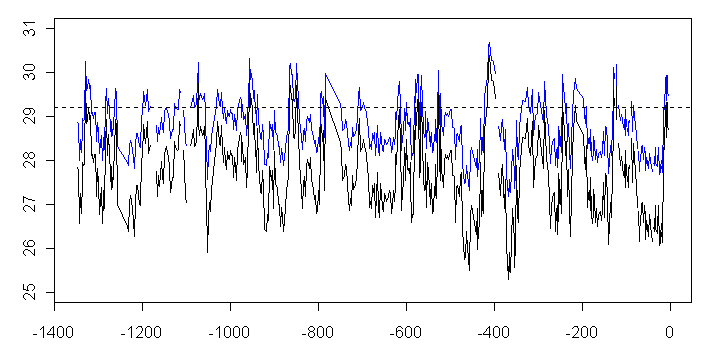
Figure 6. Hole ODP806B Reconstruction using varied transfer function. Blue – amended calculation.
Conclusion:
Where does this leave us? In paleoclimate terms, using the adjustment for dissolution as calculated above, the temperature differences in the Pacific Warm Pool are less than one would expect. My guess is that the dissolution adjustment in glacial times will be less than in warm times – there’s substantial evidence at modern sites that dissolution is not as serious a problem at cooler sites. However, as long as one is speculating on the proportion of dissolution in the foraminifera, there are layers of uncertainties that are not even hinted at in Hansen et al 2006. We don’t know what the pre-dissolution values of Mg/Ca for the Holocene Optimum were and so comparing modern instrumental values to core top values is little more than speculation. Having said that, Mg/Ca levels in the Holocene Optimum were relatively warm within the Pleistocene – reinforcing the observation that the Holocene is a relatively mild period within the Pleistocene. Prior to the last few years, this was usually believed to be a “good thing” in human terms.
For calibration, the Barker et al 2005 equation pertains to calcification temperature. G ruber grows in the mixed layer – I’ve seen a figure of 0-50 m quoted in the South China Sea. Presumably it would grow deeper in the WEP with its very deep mixed layer. So the temperature that’s being measured is some sort of average temperature in the mixed layer; how does this compare with the instrumental measurement? Well, consider all the energy that went into adjusting between canvas and wooden buckets in order to make the HadSST data set. If those adjustments are worth making, then surely a little inquiry is required before splicing calcification temperature integrated over the mixed layer in some manner with instrumental temperatures.
Update (Oct. 2, 2006)
In some comments, I pointed out that there was evidence [McClain et al,. JGR 1999] that the top part of the Warm Pool was nutrient poor and that plankton growth optimized at lower depth, which presumably would be cooler by about 2 deg C. David Stockwell posited that the unexplained 1.6 deg C adjustment in Dekens et al 2002 plausibly came from this. Here’s some documentation of this effect and a diagram of its impact.
Tian et al 2005 say of the South China Sea:
G. ruber is a mixed layer dweller that lives at depths between ~30–60 m in the upper mixed layer of the modern ocean [Hemleben et al., 1989],
Peeters et al 2002 say of the Arabian Sea:
The calcification temperatures of G. ruber mirrored the seawater temperatures near the DCM, at 11 m during upwelling (at station 313). Although calcification temperatures during non-upwelling ranged between the sea surface temperatures and those found at 80 m, the average calcification temperatures suggest that most calcite precipitated between 50 and 80 m, i.e. between the DCM and the upper thermocline. On average, the calcification temperature of G. ruber was 1.7 deg C lower than the sea surface temperature.
These admittedly do not specifically cover the Pacific Warm Pool. McClain et al 1999 discuss the Pacific Warm Pool and, while they do not specifically discuss G. ruber, they have information that bears on the problem. They report on model output for plankton production in the Warm Pool at 165E, not too far from the Ontong Java Plateau. Here is a graphic showing levels of plankton productivity:
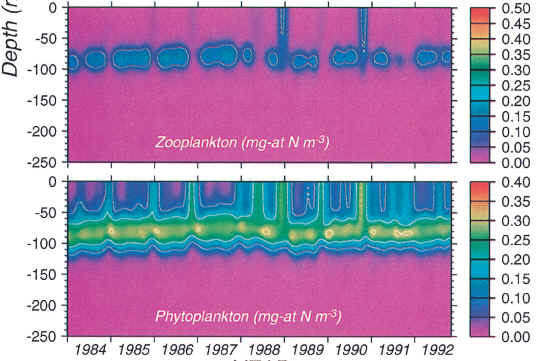
Plate 5. Depth-time contour plots for simulated (a) NO3 2, (b) NH4 1, (c) Z, and (d) P (or chlorophyll) in mgatN m23 derived from the diffusion-only (w 5 0) simulation. McCl;ain et al JGR 1999.
They also provide the following graphic showing the temperature output from their model while noting in their text that
“The temperatures replicate the observed temperatures within the upper 100 m to within ~1 deg C but are generally warmer at depth by 1–2 deg C, on average. (p 18311) “
.
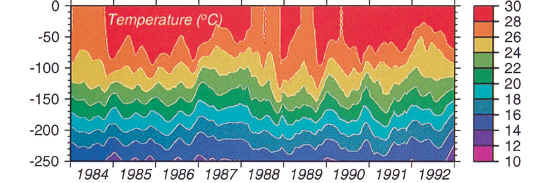
Plate 2. Depth-time contour plot of … (d) temperature (8C) from the ocean general circulation model. Texr:
Thus, as David Stockwell suggests, it seems quite plausible that the adjustment of 1.6 deg C in the Dekens et al 2002 equation describes the difference between SST and the average calcification temperature for G. ruber. If so, then the above equations should read, allowing for the algebraic
(10)
This produces the following rather troubling graphic:
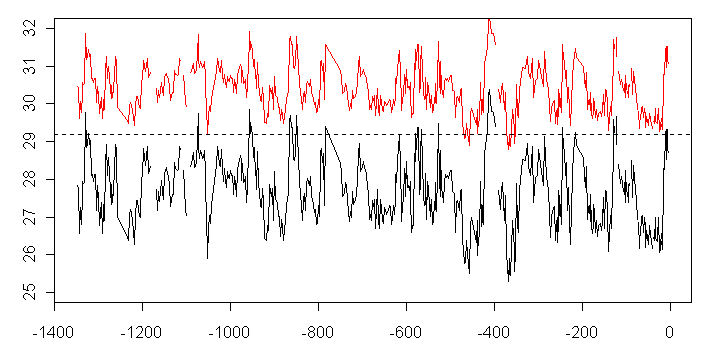
Figure x. SST Reconstruction using equation (10), which assumes that the Mg/Ca reconstruction estimates a mixed layer that is 1.6 deg C cooler than the surface SST.
I think that what this latest calculation is saying is that a linear adjustment for relative dissolution is probably not very accurate, but it obviously highlights the impact of assumptions on Mg/Ca dissolution in trying to carry out a Hansen splice.
Data Digression
Now for a little digression on the data in this diagram. As usual in matters paleoclimate, it’s never easy to reconcile data and I’ve spent a couple of days trying to sort this out. Lea et al 2000 Table 1 and Dekens et al 2002 Table 4 (both shown below) show core top Mg/Ca and SST for a variety of cores, including cores shown here. Lea has archived a couple of holes (notably ODP806B and TR163-19 at WDCP – excerpts below). The trouble is that the data does not reconcile exactly and doesn’t reconcile clearly with the figure. Now it’s close enough that probably not much turns on it, but it’s very frustrating when you’re trying to figure out what they did.
For example, the left panel of Lea et al 2000 Figure 2 has two points at 26.2 deg C with Mg/Ca values just below 3. These cores are almost certainly TR163-18 and TR163-19, but the values in Lea et al 2000 Table 1 are a little bit higher than in Figure 2. The values for TR163-18 in Dekens et al 2002 are different: these might be the values used in Lea et al 2000, but what is in Table 1? What is the connection between the archived values and the reported core top values? They are close in both cases – but don’t match. I’ve experimented with different combinations but haven’t been able to determine what was done. Other puzzling points: why is TR163-22 shown in Lea et al 2000 Table 1 not carried forward to Dekens et al 2002 Table 4? However aside from these questions, the cores used for the fitting appear almost certainly to be (from left to right) the following 11 cores: TR163-32; TR163-27; TR163-28; TR163-20B;TR163-22; TR163-18; TR163-19; ODP806B; MW91-9 08; MW91-9 34; MW91-9 38.
Excerpt from Lea et al 2002 – Table 1


Excerpt from Dekens et al 2002 Table 4
Excerpt from Table S2 Medina-Elizalde and Lea 2005 SI. http://www.sciencemag.org/cgi/content/full/1115933/DC1 for ODP806B (also at WDCP here . For data on TR163-19 see WDCP here
References:
Lea et al 2000. Science
Dekens et al 2002. Paleoceanography.
Barker et al 2005. QSR.
Hansen et al 2006. PNAS.
Tian et al 2005 GRL
Frank J.C. Peeters, Geert-Jan A. Brummer, Gerald Ganssen, 2002, The effect of upwelling on the distribution and stable isotope composition of Globigerina bulloides and Globigerinoides ruber (planktic foraminifera) in modern surface waters of the NW Arabian Sea Global and Planetary Change 34 (2002) 269–291
S. Levitus and T. P. Boyer, World Ocean Atlas 1994, Volume 4: Temperature, NOAA Atlas NESDIS (U.S. Department of Commerce, Washington, DC, 1994).Accessed at http://ingrid.ldgo.columbia.edu/SOURCES/.LEVITUS94/




25 Comments
The way I read the Fig 3a caption, they are correlating Mg/Ca measurements on the sea floor (core-top) at depths of 1625 to 3200 m with the SST. That seems like quite an assumption in itself.
The measurements on the sea floor are of a foraminifera (G ruber) that lives in the mixed layer and which have “rained” down to the sea floor. So the material did originate at surface and I don’t see that as problematic in itself.
Excellent post, Steve. Easy to understand and you do a very good job of highlighting the problem in the studies.
I am stunned at the same things you seem to be (judging by the !?! you added) – the circular reasoning that is implied here. That is, formulas are calibrated using Holoscene Optimum proxies and modern SSTs. If Holoscene Optimum temperatures are higher than modern SSTs, this is going to bias the formula downwards. Then they use these formulas to calculate Holoscene Optimum SST (biased downwards) and then act shocked when the results are lower than modern SSTs!
Ignoring all the rest of the uncertainties (where are the error bars on Hansen’s graph???) this is a sad indictment of the circular reasoning which seems to integral to climate field studies today.
I’m once again amazed at what one finds in studies once one digs into them. I’m also impressed with how well you analyse these studies and how carefully you check the data.
Steve,
Given the spread on the exponential fit shown in Figure 5 (seems like the better part of +/-2.5 C at most molar rations) and rather poor fit of the exponential curve to the data at the end points, how does one get a +/- 1 C confidence?
A few questions to think about:
1. The sediment trap data (Fig. 5) are the most accurate correlations of Mg/Ca values to SST because the forams will have been collected shortly after they expired and began to fall to the seabed. The spread on the data points overall and for individual species looks rather large so the question is why. How do the shells get transported downward? I recall reading something long ago about the fecal pellets of predators being a mechanism. Do others fall as solitary shells and what impact on Mg/Ca does this have?
2. Dissolution is related to the carbonate compesation depth, sedimentation rates, and bioturbation. Have these been factored into the equations satisfactorily?
3. Lab processing of samples is difficult and you don’t always get good pickings. The level of experience and training may be impacting some of these measurements, expecially with a new method. Likewise, the siting of deepsea cores is not always for the purpose at hand – maybe they were really after basement rock at the bottom of the sediment and the microfossils are an “extra”. Sometime all you can get is close. This doesn’t excuse sloppy work or failure to adequately document less than perfect samples, however.
So you are saying:
1. Hansen used Lea’s superceeded equations that did not adjust for dissolution of Mg at depth.
2. The most recent eqns for adjusting for dissolution contain a serious (and trivial) algebraic error.
3. When results are corrected for dissolution with correct equation, mean of reconstruction increases by up to 2C and SD halves.
I am amazed but not surprised. Thanks again Steve.
Now I can hear them saying already, but it makes no difference, its still the warmest in a millyun years.
David, plankton in the PAcific Warm Pool is a nice ecological niche situation. I’ve been reading somne more. It seems (McClain et al , JGR 1999) that there is a nutrient-poor zone at the top of the warm pool; thus plankton growth reaches a maximum between the surface and where light is attenuated. I’ve not located a clear diagram showing temperature gradients but a diagram in the same paper suggests that temperatures at the plankton maximum are about 2 deg C cooler than the surface Warm Pool. For Lea’s original article in which his interest was interglacial differences, this would probably not be relevant, but if you’re trying to do a Hansen splice, it obviously is.
As to the likely error in Dekens et al, it’s possible that glacial climates had lower dissolution rates and that this would mitigate the changes from fixing the algebraic error.
There have been a lot of articles on dissolution in the past 6 years. It’s hard to understand why Medine-Elizalde and Lea re-used the old equation in their 2005 article. I’ve sent an inquiry about this to Lea – I wonder whether I’ll get an answer.
Is this the source of the 1.6C constant in the calibration equation of Dekens but not in yours?
If dissolution occurs at depth then surface temps wouldn’t be such a factor.
#8. 1.6 deg C – That makes a lot of sense although they didn’t mention it. I’ll try it that way and update the note.
David, your explication of the 1.6 deg C adjustment made even more sense when I checked on it. Peeters et al (see quote in update) explicitly state that calcification temperature for G ruber in Arabian Sea is 1.7 deg C cooler than SST; admittedly that’s not the Warm Pool, but I’ve shown related info from the Warm Pool to show that plankton growth maximizes at lower depths and that those lower depths are cooler. A 1.6 deg C adjustment between G ruber temperature and SST seems quite reasonable for Dekens et al to have done.
Howeever that causes even worse havoc with their reconstruction when the algebra is corrected -see the chart. All this does is throw into sharp relief the problems with guessing at how much Mg/Ca dissolution has taken place. Based on the information presented in Hansen et al 2006 and his sources, it’s not possible to even make a wild-assed guess on the crucial parameter. The more one digs into this, the worse it looks.
#10. This obviously needs a fine-tooth comb. It looks to me that the Mg/Ca ratio is linear with water depth not an exponential function as shown by Lea’s RH panel. So Dekens equation must be wrong. But Lea’s by omitting known dissolution effects are also a problem.
1. Are you sure that the direct calibration of Mg/Ca ratios with SST doesn’t result in incorporation of the optimal growth of G. ruber at 1.7C below SST?
2. There is a big difference between the postion of G ruber on the LHS Figure 3 and G. ruber on Figure 5. The coefficients are different too – 0.3 vs 0.38 and 0.9 vs 0.89.
#11. 2. THe scale of the two RH panels is different – my emulation shows a greater depth than in Figure 3, but allowing for scale the emulation is (I think) correct.
#1. I don’t vouch for my interpretation. I’m obviously learning as I go and will correct any misunderstandings. I think that the linear fit in the Lea et al 2000 right hand panel may be part of the problem. THe actual result is not at all linear since Mg/Ca ratios are ~0 by 3500 m.
Here’s another online reference:
Pallavi Anand and Henry Elderfield, Maureen H. Conte, 2003. Calibration of Mg/Ca thermometry in planktonic foraminifera from a sediment trap time series, Paleoceanography. http://www.geo.vu.nl/~palmorph/staff/pallavi/Anand%202003%20Paleo.pdf
#12. Yeah but it shouldn’t be as it seems pretty basic. What I meant by the difference in the postion of G. ruber in the LHS panels is that it seems like the temperature is translated by a few degrees. That is the calibration of Lea puts the G. ruber niche a few degrees warmer than the calibration graph of Barker. I get the impression that Lea is a direct calibration to SST while Barker is a calibration to temperature at collection depth. Is that right?
Isn’t there some chemistry behind this?
The "empirical" Lea calibration relates Mg/Ca in the low 4s with Warm Pool temperatures and I suppose is a "direct" calibration. Such a calibration, in a sense, includes the impact of both dissolution and calcification depth without dealing with either issue. For relative changes, it might be reasonable enough – it’s only when Hansen tries to splice it with the instrumental record that he goes a bridge too far. In order to splice, it seems to me that he has to have control of all the factors – but none of them are known to the degree that he requires.
#14 While Lea’s calibration may account for calcification depth it wouldn’t account for dissolution would it, even if its direct? That is the samples are collected at different depth sites and this is uncontrolled. Its only a few points, and just by eyeballing it, I wouldn’t think exponential was a very appropriate fit anyway — looks to increase more quickly than that. Well I would linearize it anyway and work with the linear function forms but that BTP.
Whether or not Lea is comparing Holocene Mg/Ca with modern temperatures depends on the exact meaning of “coretop”. I was assuming that they separately sampled the very top of the ocean floor in the area, not the part a couple of hundred mm down which is the start of their Holocene record … but they may actually mean the top of the core, which would be odd.
w..
I saw a reference that dated the “core tops” to 4000-6000 BP, but will have to go back for the citation. It doesn’t make any sense that the core top actually is from the Holocene Optimum – why wouldn’t there be any core for the last 4000 years, although there have been missing intervals from time to time in the past. The most detailed records are at WDCP – see the link in Data Digression.
The control on dissolution appears to be the pCO2 value as discussed in Dekens. But they don’t know this column and depth is a “proxy” for pCO2.
RE: Plate 2 – the cool spikes got me to thinking / asking. Just how close is that site to the Strait of Malacca / Indian Ocean? The reason I ask is, in that area, the Indian Ocean is cooler than the Pacific. Is there an interaction between the two oceans impacting the data?
1. I know nothing about this stuff, but I’m curious: Wouldn’t the dissolution rates of Mg, Ca depend on temperature itself? Or is their dependence identical? Has this been accounted for? Would that bias the temperature readings in one direction or another? Even if the difference in temp dependence is minor, over time could this be significant?
2. BTW, without getting into religion, y’all should adopt the motto supposedly enshrined in a plaque at NASA’s Johnson Space Center: “In God we trust. All else bring data.”
re: #19
Well, Calcium carbonate has a solubility in cold water of about .0015 while Magnesium carbonate has a solubility of .17 or roughly 100 times as great. This is partly reflected in their relative concentrations in seawater on a molar basic where were magnesium is 6 times as common. But that still means magnesium would be more likely to dissolve than calcium, other things being equal. Though I notice there’s one form of Magnesium carbonate which isn’t nearly as soluble as the plain hydrate is. Still, as time passed I’d expect the ratio of magnesium to calcium to decline.
But as to the kinetics of the situation, I’m not positive. I don’t seem to recall the thermodynamic equations which govern such things making a distinction between different compounds. But there are lots of imponderables unless you look at a particular situation. There might be a different rate-limiting step which would favor one compound’s dissolving over another. And the crystalline form could also be important. And are the compounds co-mingled or do they form separate crystals?
I don’t know if dolomitisation should be considered here, but as it is CaMg(CO3)2 here is a discussion how its formed:
Click to access 07_Chapter1.pdf
from
http://www.jcdeelman.demon.nl/dolomite/bookprospectus.html
MORE BRACKETS …
Re 8, 9:
This is not the case. The 1.6° constant is to adjust for a difference between the Atlantic and the Pacific. Dekens says:
The resulting equation is:
Mg/Ca = 0.38 exp (0.09*(SST -0.61*core depth km))
for G. ruber, Atlantic and
Mg/Ca = 0.38 exp (0.09*(SST -0.61*core depth km) – 1.6 C ))
for G. ruber, Pacific …
However, this is related to another problem. Steve M., you say,
Actually, Dekens has done something quite clever. He has done a multiple regression of the form
where Y is SST [Levitus and Boyer, 1994], X1 is ln(Mg/Ca), X2 is water depth of sediment (km), and X3 is 0 if the core is in the Atlantic or 1 if the core is in the Pacific. Regressing this gives coefficients that match the data best. Investigation showed that the b3X1X2 term was very small, so he was left (for the Pacific) with
This resolves to the formula he gave. It appears that the similarity between the 0.61 and the 0.58 was coincidental.
However, there’s more to the story. I examined the Figure 5 you showed above:
I noticed that there’s a whole raft of species in there, whereas Lea only used G. ruber (white). So I went back to the Anand study referred to in Figure 5. Anand breaks it down into spinous species, which includes G. Ruber, and non-spinous species. His formula for the spinous species is
Mg/Ca = 0.58 e (0.075 * SST)
Note the difference between that and the formula for all species, with coefficients 0.38 and 0.9 respectively.
I took the G. ruber (white) data alone, and regressed the best match for that. Here are the results:
Note that the Lea and the Dekens results are very similar. This may be why, despite the advance, Lea continued to use his formula. However, neither one is a very good match to the G. ruber data. I show the best match. I then took the best match, and regressed it on the Dekens formula to find the correct depth adjustment. Using that formula gives the following results on the Lea data.
Note that the Dekens and Lea data are quite close. The Best Match results reflect the slightly different slope due to the improved match with the G. ruber data alone, and thus have a slightly wider spread. In this regard, it is worth noting that Lea commented that his results did not show as wide a spread as expected, saying
Finally, a conundrum. I looked at the sedimentation rate at the OJP806 site. Here’s Lea’s figures:
Does that make sense to you? Look at the area about 250,000 years ago. Is it reasonable that the sedimentation rate would stay absolutely stable (within the limits of resolution) for tens of thousands of years, and then increase threefold in only 800 years? I don’t think so … but what do I know?
w.
#22 That’s great Willis. Mine was a case of ready-fire-aim. Your improved calibration seems to show that both early holocene and 100kyrBP temperatures could have been higher than the present, plus a whole raft of peaks, and make a significant impact on the Hanson claim.
#22. Willis, I agree with your reading of Dekens and will revise my comments. I couldn’t get the numbers to work when I tried a regression using the data in Dekens. Do you have your scirpt handy so that I can reconcile?
#24, Steve M., sorry, no script, for most things I use Excel because by the time I could get the R script written, I have the results and graphs done in Excel and have written the post … plus I have better control over the graphics. R is wonderful, don’t get me wrong, and I wish I knew it better, but that’s how it is. I have a number of special-purpose functions I’ve written in Excel, and can drive it very fast, so I use it most of the time.
Also … thanks for putting together and riding herd on this wonderful blog.
w.